Normality Calculator
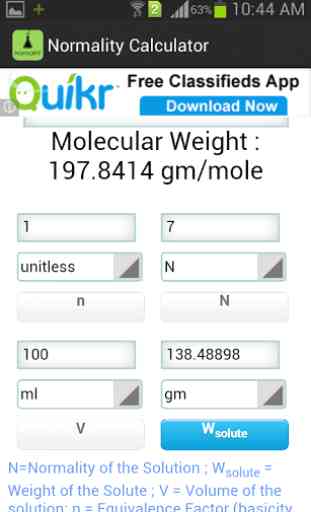
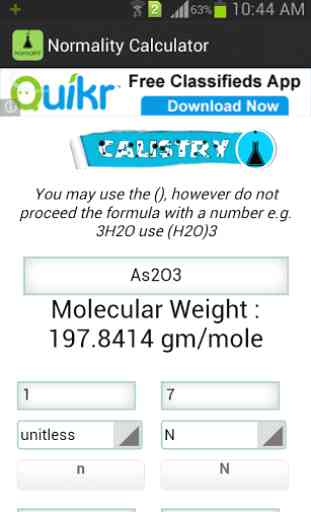
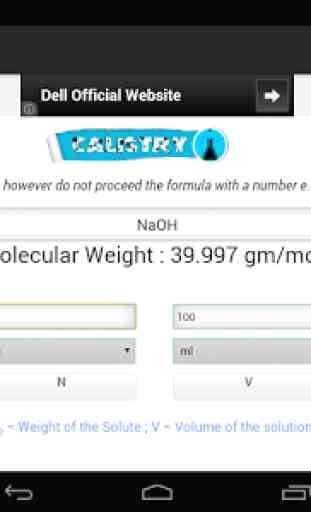
Use this calculator for preparing a solution of solid solute. To use the calculator , first enter the formula of the solute. Then enter any of the three of the four parameters, namely: N=Normality of the Solution ; Wsolute = Weight of the Solute ; V = Volume of the solution; n = Equivalence Factor (basicity of an acid or acidity of a base or number electron transfer in a reaction)
Description :Normality of a solution is defined as the number of gram equivalent of a solute present per liter of a solution. The gram equivalent is the ratio of molecular weight and equivalence factor. This equivalence factor can be thought as the number of replaceable H+ ions in a molecule of an acidic solute or the number of replaceable OH
- ions present in a molecule of a basic solute. Thus equivalence factor of NaOH is 1 and 2 for H2SO4. In the case of redox reactions this equivalence factor is the number of electron taken or given out of that compound in a redox reaction.
This calculator is the android version of the following web application. Visit: http://calistry.org/normalityCalculator
Description :Normality of a solution is defined as the number of gram equivalent of a solute present per liter of a solution. The gram equivalent is the ratio of molecular weight and equivalence factor. This equivalence factor can be thought as the number of replaceable H+ ions in a molecule of an acidic solute or the number of replaceable OH
- ions present in a molecule of a basic solute. Thus equivalence factor of NaOH is 1 and 2 for H2SO4. In the case of redox reactions this equivalence factor is the number of electron taken or given out of that compound in a redox reaction.
This calculator is the android version of the following web application. Visit: http://calistry.org/normalityCalculator
Category : Education

Related searches
Reviews (4)
VΞɴ. S. C.
Oct 14, 2014
It is not an normal Normality app
Sup. M. A.
Oct 10, 2014
liked it very much
Tar. G.
Oct 5, 2014
Help full

Loved it